APPROACH: WHERE IS THE LESION, WHAT IS THE LESION?
History - how to localise with questions
We approach neurology the same way we do any area of medicine - first the history, then the examination, and base our diagnostic impression on these, then investigate to confirm or refute one or more hypotheses. Investigations should be tailored to the situation and we should have an idea of how we will react to positive, negative or ambiguous results.
Perhaps more than any other branch of medicine, neurology depends heavily on the history, and we use it to do most of the work needed to answer our two questions. Good history-taking is arguably the major skill of neurologists.
In practice we do not structure our history into ‘where’ and ‘what’ - we elicit both naturally through listening and asking. For illustrative reasons I have separated the two questions out to show how the history answers both.
This is not a complete guide to history-taking, and there is no single ‘structured neurological history’ - it varies with the problem. This is a specific focus on ‘where’ and ‘what’, with practical suggestions on how to explore them, including how to clarify what patients are really saying - rather than taking things at face value.
There is a very long list of neurological symptoms - I've listed some in the Definitions section. Some localise to quite narrow territories - such as monocular visual loss. Others can arise from a wide range of sites - such as difficulty using a hand.
The history tells us what the symptoms are, and as we take it we should be aiming to map the symptoms to a given region of the nervous system - initially in broad terms (i.e. the rough ‘neighbourhood’) and then fine-tuning.
The crude localisation may be obvious immediately, for example a headache (nearly always cranial region), cognitive disorder (brain), or vertigo (vestibular system, brainstem or cerebellum).
However for other symptoms there are multiple possibilities, particularly if a single limb is symptomatic - for example, unilateral distal leg weakness could be a very distal lesion in the lower motor neuron (e.g. common peroneal nerve), a very proximal upper motor neuron lesion (motor cortex), or anywhere in between - so the localisation is broad.
Mapping symptoms to a siteWe map the symptoms by asking questions to elicit such features as the following.
1. DistributionDiffuse symptoms can ironically be easier to localise than those in a narrow region - if the cause is a single focal lesion. If symptoms affect the whole body on one side, the problem is in the opposite hemisphere. In contrast, symptoms limited to a narrow site (e.g. foot drop) could potentially arise from a very wide number of sites in contrast and are harder to localise without looking for additional clues that narrow it down (e.g. sensory loss in a given distribution).
2. LateralisationIs the issue on one side, or both? If it affects both sides in different areas (e.g. right face and left body), or in different modalities (e.g. weak right side, numb left) this has high localising value (unilateral brainstem or spinal cord respectively). Bilateral symptoms are usually not due to brain pathology - but narrowing down further takes detailed assessment.
3. Symptoms indicating a lesion in a specific regionExamples include cognitive problems or seizures (brain), sphincters (spine or cauda equina), and fatiguability (neuromuscular junction). These are useful clues to look for.
Listening and clarifyingWilliam Osler said:
'Listen to the patient - they are telling you the diagnosis'He was correct, though we aren't passive in this process; we should always be listening, including as we ask questions to clarify what is being said.
The history is best done by letting the patient speak for themselves, and listening carefully - not just to what they say, but how they say it, and what was their reason for seeking medical attention. A huge amount can be gathered from the latter, particularly if it wasn’t their own idea to consult a neurologist. If they brought someone along, observe them while the patient speaks - body language gives away a great deal. If the history involves cognitive changes or a period of altered consciousness, take a collateral history - a crucial skill.
For symptoms such as weakness, clumsiness or sensory disturbance, get a sense of the distribution while the patient speaks. They may state this clearly (‘My hands don’t work’). They may even demonstrate affected areas (e.g. circling a numb patch on the thigh). They may also not clearly state them (‘I’ve got weak legs’), but speak of their functional impact, such as reduced ability to walk a given distance, or a tendency to falls.
After this, we then interrogate for details, including carefully exploring language used - words stated often mean something else, so don’t take them at face value. This is a natural part of how people communicate about their symptoms. Often the subjective experience of a symptom feels quite different to what is actually happening. ‘Numbness’ frequently means ‘weakness’ and vice versa. ‘Dizzy’ can mean any number of things and often doesn’t mean dizziness at all. ‘Seizure’, ‘vacant’, ‘shaky’ and ‘slurred speech’ are other examples.
If medical terminology is used, clarify what is meant by it. This can cause offence if done clumsily, but we should not assume the term is being used properly - many reported ‘tonic-clonics’ are neither tonic nor clonic and often are not even seizures.
It’s best not be blunt - e.g. ‘What do you mean by dizzy?’. This can sound aggressive; the patient is not on trial, and they should be treated with dignity.
Better to ask them to expand - ‘Can you describe the feeling in more detail - what exactly are you experiencing?’. Try not to put words in the patient’s mouth (‘Would you say it felt like the room was violently spinning?’). Avoid leading questions (‘You didn’t have any slurred speech, did you?’).
Screening for other featuresWe should then ask about other symptoms not volunteered - i.e. the systemic enquiry. Such symptoms may have been subtle, or not taken on much significance in the patient's mind relative to the ones they volunteered, but they may be very relevant to making a diagnosis.
There are too many neurological symptoms to fully cover. It’s best to focus on those relevant to the presentation which discrminate between causes - e.g. for vertigo, other symptoms related to hearing (e.g. tinnitus) or posterior fossa structures (slurred speech, double vision, clumsiness).
This part is somewhat ‘checklist’ - ask specific closed questions (‘Have you had any double vision?’). Avoid multi-option questions (‘Have you had any clumsiness, slurred speech, or double vision?’).
There is a risk that suggestible patients will answer yes to symptoms they haven’t had - so don’t take a ‘yes’ at face value. Ask for more detail, such as examples of when the symptom has happened and what they’ve experienced.
The person accompanying may be able to comment - if they’ve noted the symptom too, that’s valuable and lends it some weight. In some situations they’ve noticed the symptom while the patient has not, which may in some cases indicate cognitive issues - or an concerened relative over-reporting things. This is common in stroke-related symptoms such as facial asymmetry or slurred speech, which the public have been trained to be vigilant towards.
Functional consequencesAsk about the functional effects of symptoms. This is important to get a sense of how severe they are and if the patient is at risk, whether of falls, work performance issues, or inability managing at home without help.
It also offers localising value - certain functions are affected by particular neurological problems:
It is also useful to work out what patients are no longer able to do - or what they’re avoiding doing. They may not report too many difficulties in daily life, but that’s because they’ve stopped doing things they used to - ask what those were, if they noticed problems during them, and what would happen if they tried now. People who are functioning reasonably well in daily life as a result of giving up their former activities may be significantly impaired.
We already reviewed how the different types of lesions manifest (see Where?).
The following describes the approach to exploring this in the history, considering onset, evolution, transient symptoms, associated features, and how to use demographic factors as clues to the aetiology.
OnsetThe tempo of symptom onset is a fundamental point in neurology and distinguishes the type of lesion - but the history has to be done carefully. Symptoms can arise over any time frame: instantaneous, seconds, minutes, hours, days, weeks, months or years.
It is common for symptoms to be reported as sudden-onset, particularly severe ones - but many of these actually are not. When they truly are, it is significant - particularly for vascular disorders such as stroke (with focal symptoms) and subarachnoid haemorrhage (with headache). When they are not, we can think of this as pseudo-sudden onset, and we need to consider disorders that come on a little more slowly. Always clarify what is meant by 'sudden'.
Sometimes it is obvious that symptoms began suddenly, for example a witness statement that the person collapsed in the middle of an action. Another example is someone who clearly remembers abruptly losing their ability to speak and only producing incomprehensible sounds when they tried for the next 5 minutes - then things rapidly returned to normal.
Other times it can be difficult to establish the tempo of onset. Patients may report sudden-onset, but in fact the symptoms have been present for some time when we explore the background.
Why might patients report things as sudden that actually were not? Remember that people often lack full awareness of neurological symptoms (anosoagnosia) until they reach a level of significance and have consequences:
When symptoms have been progressive over a time frame, we should try to get a sense of when the patient was last in their usual state of neurological health - their functional baseline - and then map out how things have changed. Most patients don't give us a pre-made timeline - it's our job to build one.
The rate of change varies with the situation:
In addition to onset, we can tell a lot from what happens next. Diseases all behave in certain ways, and whether they progress, stablise, fluctuate, or fully disappear, is valuable information. We use it heavily in neurology.
We don’t have this luxury when seeing a patient in the emergency setting shortly after new symptoms began - and we generally can’t sit and wait to find out!
However, we do when we see people who present after a delay, or in the outpatient setting, and this is the case for many neurological encounters.
Plateau and improvement, if present, are informative.
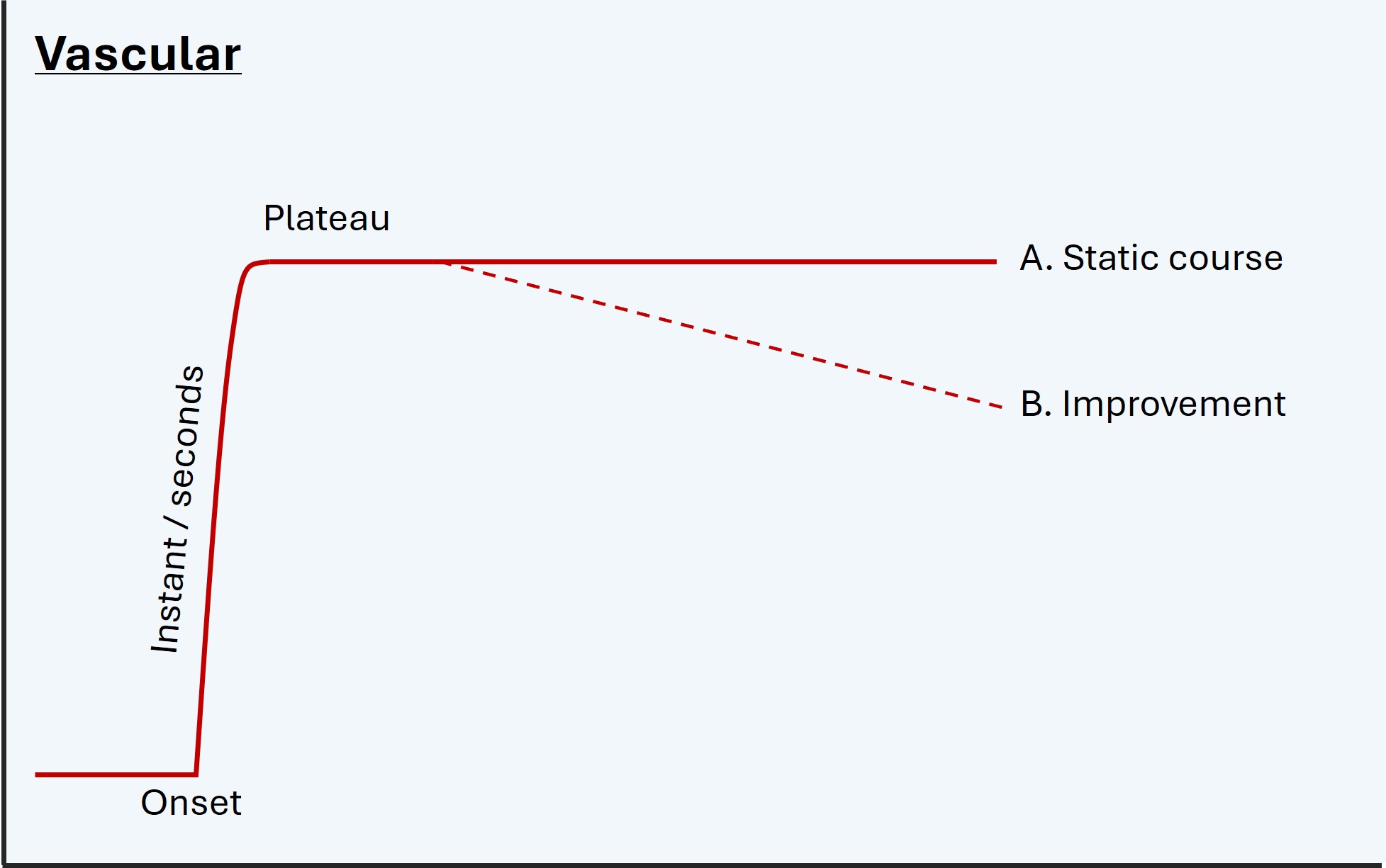
Vascular diseases come on abruptly, tend to plateau immediately or otherwise within 24-48 hours, then (if not fatal) either remain static or gradually improve - whether partially or completely - in the coming weeks and months.
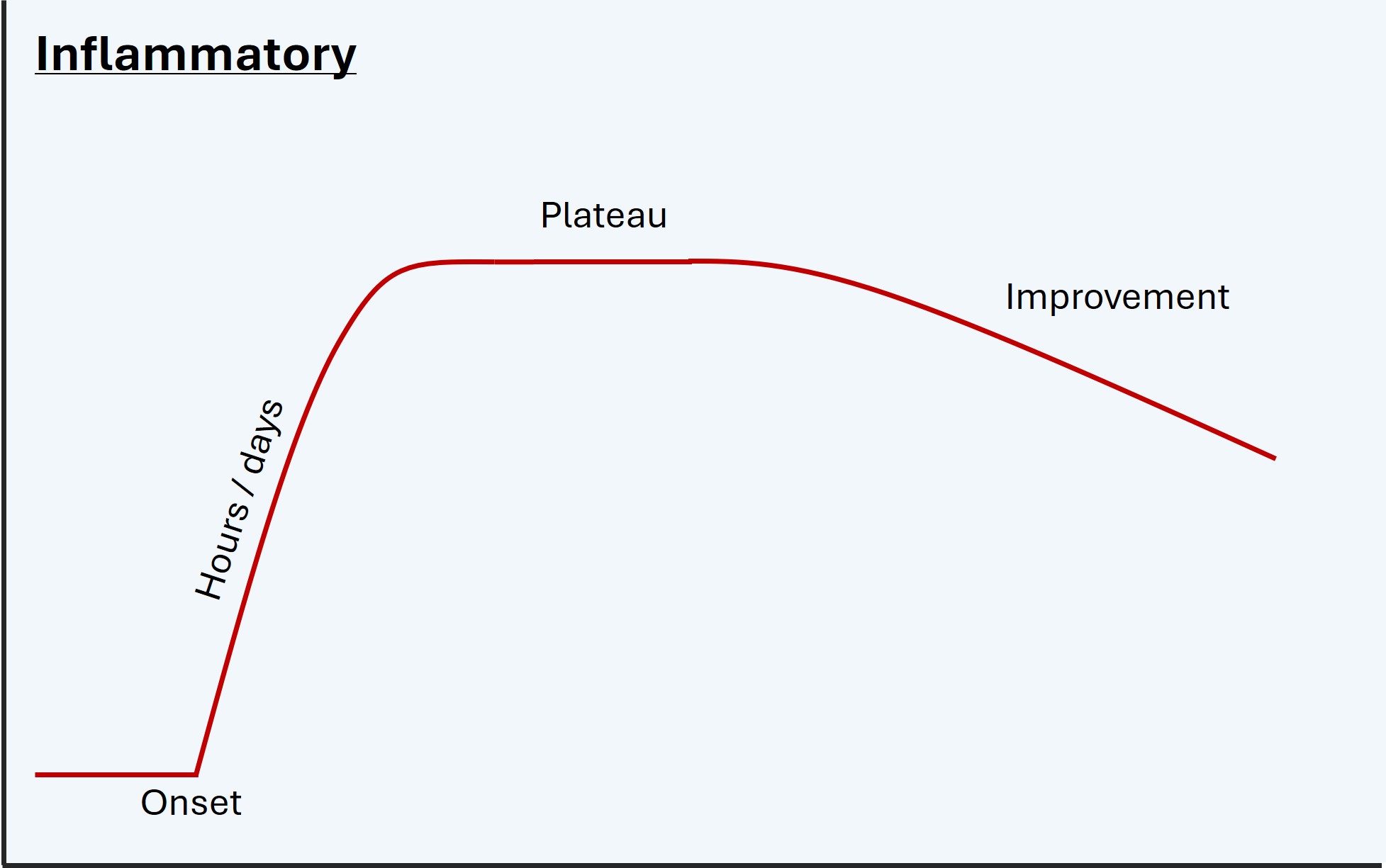
Inflammation is similar but with more gradual onset over hours or days, then a plateau, then recovering over a slower tempo (weeks-months). Examples are a demyelinating lesion causing optic neuritis or myelitis, or inflammatory neuritis of the facial nerve (Bell’s palsy). This is a generalisation though - some inflammatory disorders can progress to a life-threatening level without treatment (e.g. autoimmune encephalitis), so the natural history is not always of onset, peak, then resolution.
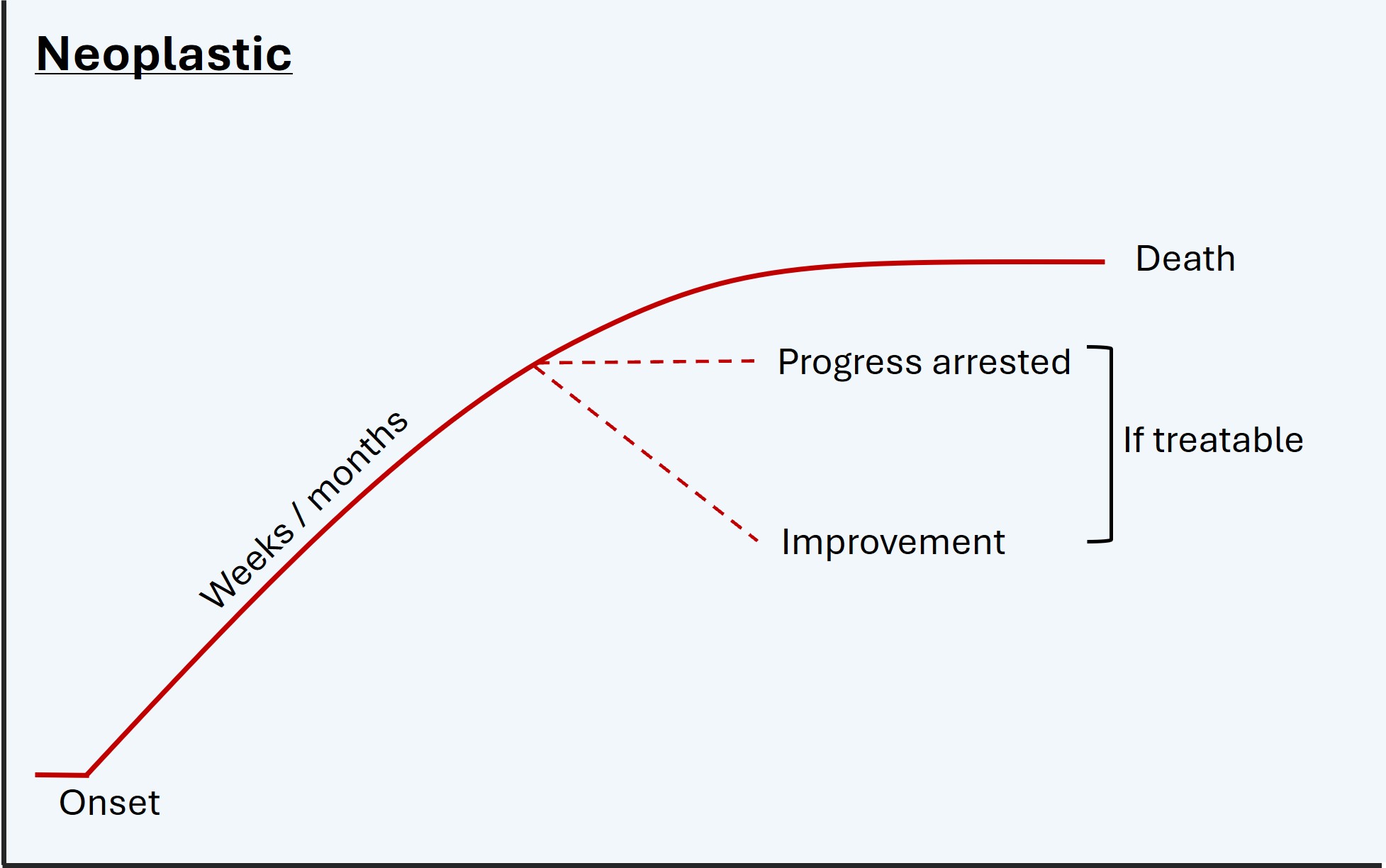
Constant worsening is also informative. Degenerative disease and untreated tumours get worse relentlessly, usually gradually over months or years. Remission does not naturally occur.
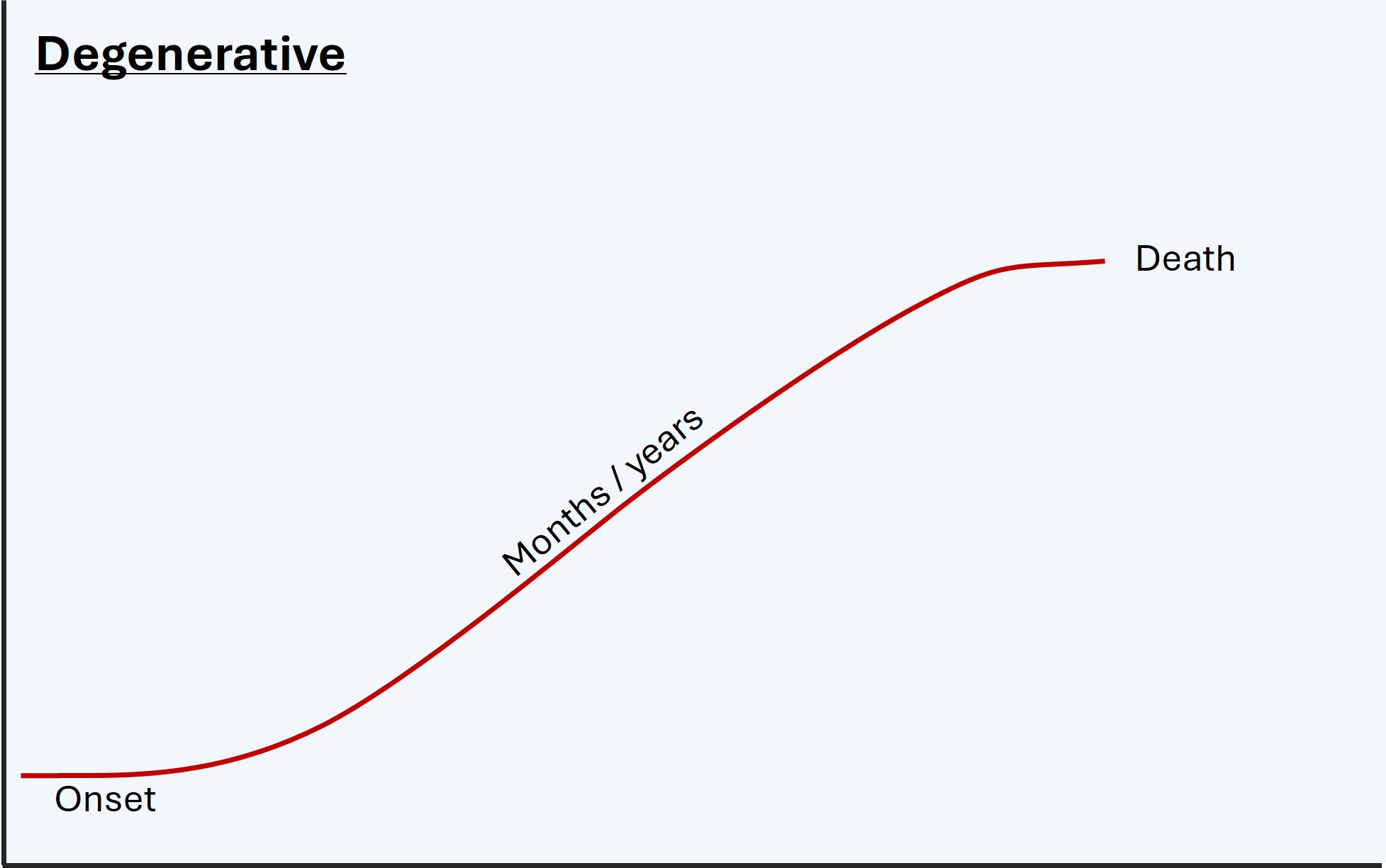
Of other types of lesion, some may naturally come on and then plateau and resolve. This is often the case with metabolic disorders, but only if the underlying issue self-corrects (e.g. hypoglycaemia) - others may not without intervention, for example encephalopathy due to renal or liver failure. Likewise, toxins may reach peak effect then be cleared from the system - the best known and most common being ethanol - although some will produce deadly effects in the peak phase, and supportive measures may be necessary to keep people alive until the toxin is cleared - for example paralysis due to botulism or organophosphates.
Onset, evolution, plateau and recovery can also be the case with compressive lesions, but usually only if the consequence is reversible inflammation in the compressed structure - the culprit lesion doesn’t tend to return to its initial position. A common example is irritation of a nerve root from a disc prolapse, which often causes severe radicular pain and sensory disturbance for several weeks and then resolves as root inflammation dies down (the disc itself does not tend to un-prolapse) - hence, surgeons tend not to operate on this in the absence of disabling physical deficits, mainly weakness.
However, other compressive conditions do not get better without surgical help, for example advanced carpal tunnel syndrome, cauda equina syndrome, or oculomotor palsy due to an adjacent aneurysm. In these, even after decompression, the damage may be permanent, and the procedure only prevents further deterioration - or in the case of the aneurysm, death from rupture.
Transient and paroxysmal symptomsA major group of disorders feature transient episodes, with symptoms happening over a short window of time. Some of these are characterised by recurrence over time and the episodes may be stereotyped, which is diagnostically very useful.
Many such disorders exist, but the ‘big three’ are migraine, ischaemia, and seizure. Features and tempos may overlap, but distinguishing features exist.
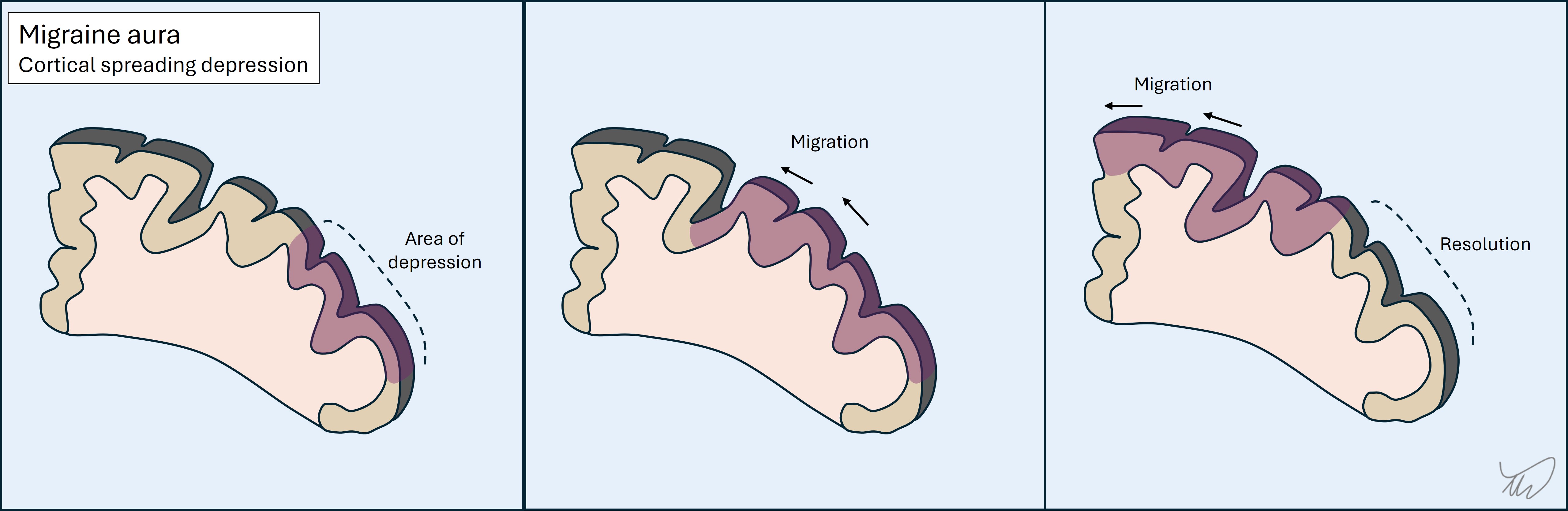
1. MigrainesThese often feature an aura, which has onset over minutes - it comes on quickly, then evolves. The commonest forms are an expanding visual obscuration or migratory tingling sensations in a limb.
Auras usually feature a mix of negative and positive symptoms, for example visual obscuration and flashing lights, or tingling and numbness together. One aura sometimes clears and another follows it, as the process migrates to an adjacent area of brain. Of note, these may move between vascular territories, which is a clue that the issue is not a TIA.
There is generally a headache following the aura(s), but not always - acephalgic migraines exist.
Migraines usually also feature other ‘migrainous symptoms’ such as nausea, light-, sound- and motion-sensitivity, and a feeling of exhaustion. As above, they are often are preceded by a vague prodrome with irritability, lethargy and difficulty concentrating.
2. TIAs
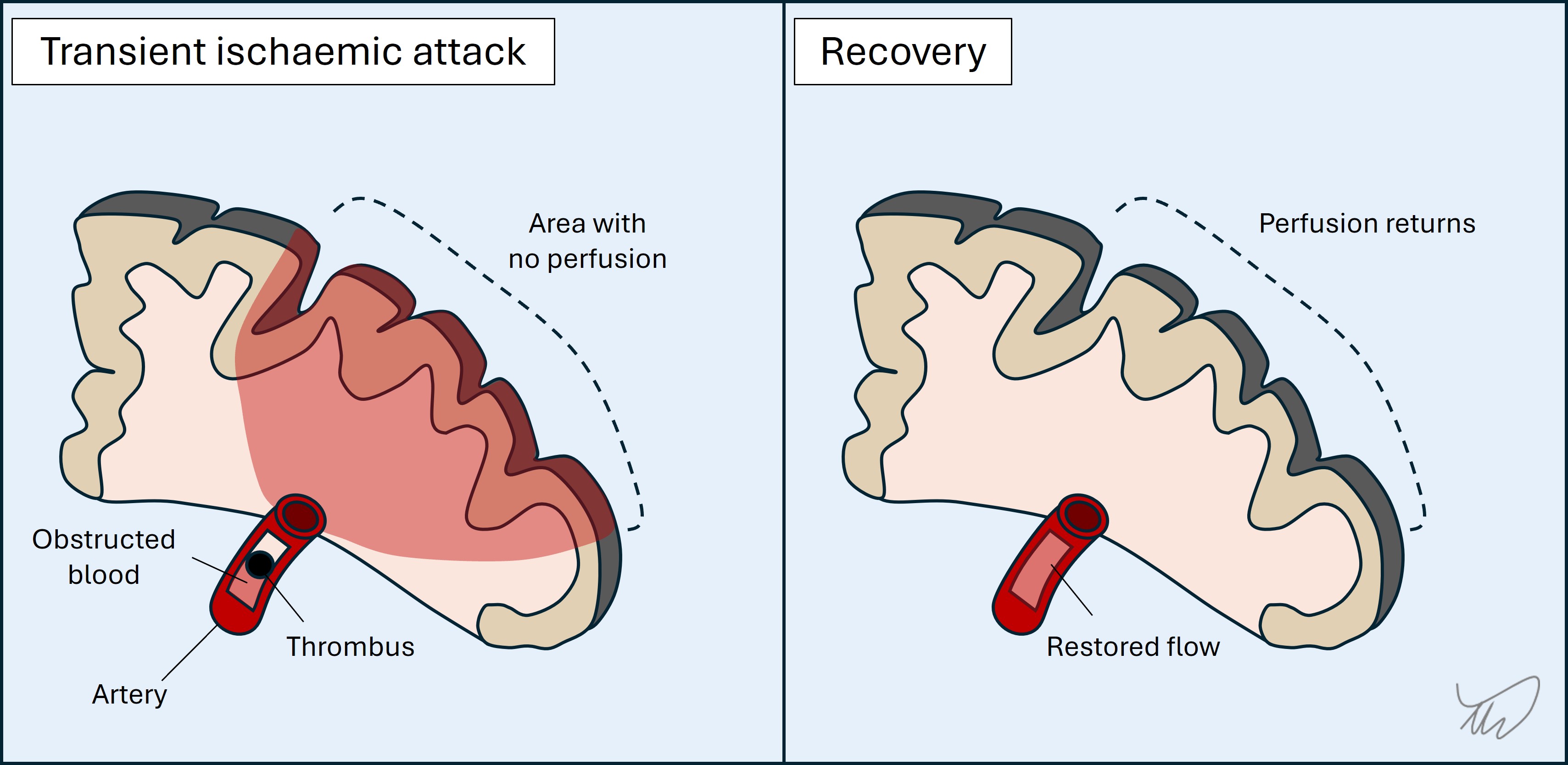
These are sudden-onset. Evolution is unusual - attacks generally reach maximal intensity and distribution at onset.
Symptoms are confined to single unilateral vascular territory (unless multiple TIAs, e.g. cardioembolic, spanning territories and sides). Symptoms may be very focal and isolated - for example dysphasia or diplopia - or more extensive, for example a combination of paralysis and dysphasia
There are usually only negative symptoms, although tingling is common in addition to numbness and is an important exception to this statement. Involuntary movements are not common nor are positive visual phenomena such as flashing lights or colours (more suggestive of migraine or occipital seizure).
Awareness is usually unaffected, although some strokes affect it, particularly thalamic ones. Basilar tip lesions can produce transient unconsciousness.
Resolution is often rapid rather than gradually ‘wearing off’.
 3.Seizures
3.Seizures
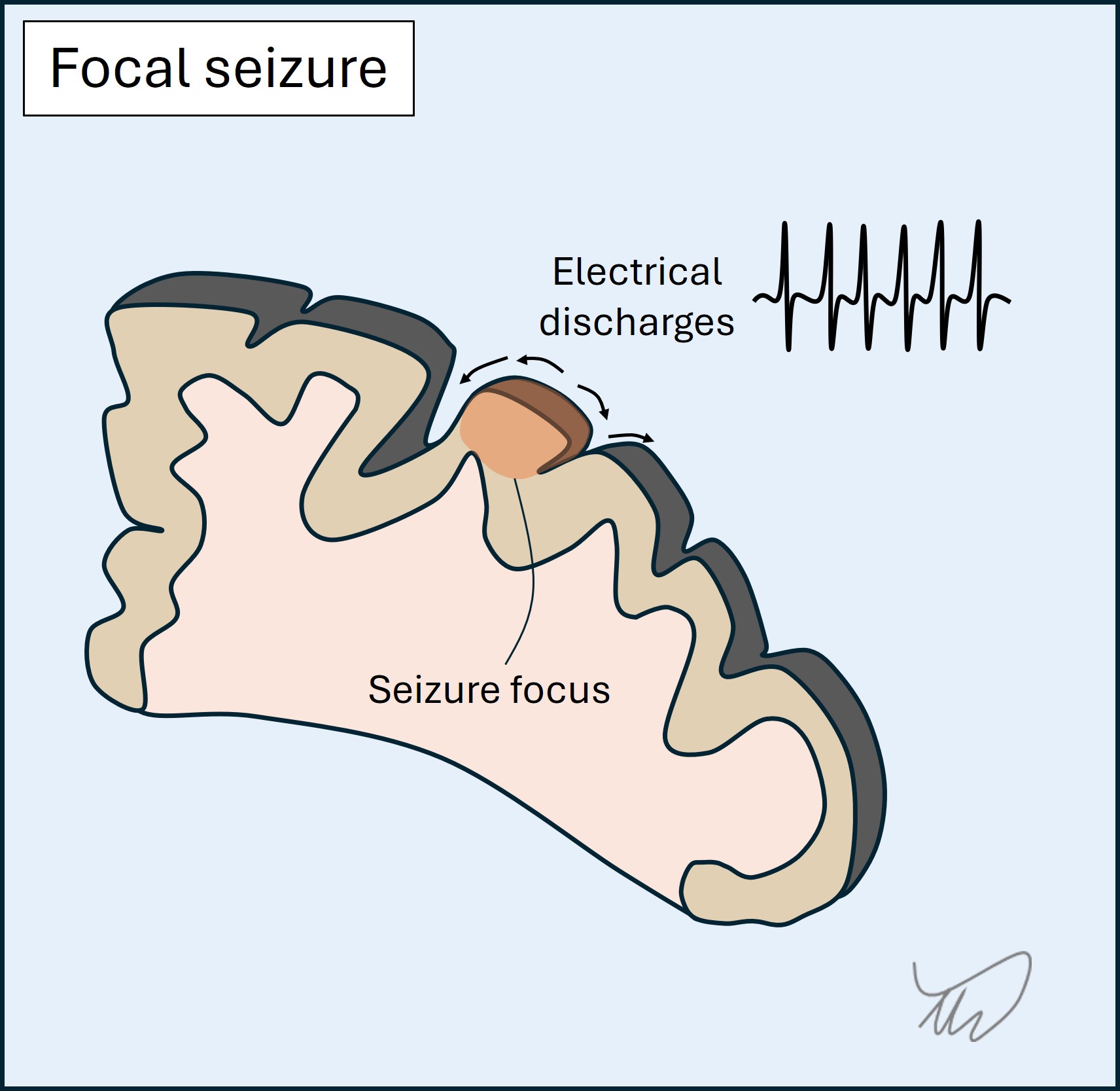
These are sudden-onset, evolve over seconds, and have short duration (usually 1-2 minutes).
They feature positive symptoms - in the case of focal seizures they reflect the area of cortex involved. The best known are jerking, abnormal sensations, and strange smells/tastes. There may also be negative symptoms such as dysphasia or numbness - but it is not common for there only to be negative symptoms.
Awareness may be affected, manifesting as vacant staring with maintained posture - people appear almost to ‘freeze’ - and sometimes with automatisms, e.g. fumbling hands, lip smacking or chewing. They may also cause collapse with unconsciousness and tonic-clonic motor activity.
A focal-onset seizure may spread to other regions, so the attack can progress through a sequence, and it may generalise - leading to tonic-clonic activity.
Finally, post-ictal deficits may arise such as weakness (Todd’s paresis) or dysphasia. In addition, a post-ictal state of confusion and lethargy (or sleepiness after a convulsion) may be seen.
A table summarising the above for the ‘big three’ is shown below; it concerns focal rather than generalised seizures.
| Migraine | TIA | Focal seizure | |
|---|---|---|---|
| Onset | Sudden but may have prodrome | Sudden | Sudden |
| Evolution | Over minutes | Usually none (maximal at onset) | Over seconds |
| Positive features | Yes, e.g. visual aura or tingling | No, except tingling | Yes, depending on site of focus |
| Negative features | Yes, e.g. scotoma, numbness, weakness | Yes - main features are negative | May be present, or follow seizure |
| Awareness | Intact | Nearly always intact | May be altered/lost |
| Aftermath | Headache with migrainous features usually | Back to normal | Confusion, lethargy, post-ictal deficits |
| Recurrence | Often, usually stereotyped | Unusual - may occur similarly over short period if a culprit artery | Yes, and stereotyped |
We rarely ever catch anybody in the midst of a brief transient episode - it is more common that we have to assess a completed one. This can be challenging. The principles above enable a clinical diagnosis - describing the symptoms, their evolution and any associated features. Doing this well is a vital skill. For the most part, no test will tell us reliably what a brief paroxysmal event was. We rely entirely on the history, and in epilepsy we sometimes attempt to catch another one ‘red handed’ during electroencephalography.
When such paroxysmal symptoms are single events, diagnosis can be tricky, and we may be forced to investigate rather than take a watch-and-wait approach (particularly for TIA). If they are recurrent this is informative, and if they recur over a long period and are similar or identical each time, this is very helpful. Seizures tend to be very stereotyped. Migraines may vary, but people often experience the same migraine (including prodrome, aura, headache and aftermath) - with experience they know when it’s coming on and what to do.
A recurrent attack happening identically over a long period with long interludes between them is not likely to be TIA - but over a shorter timeframe and with little interlude, it may indicate a critically narrowed artery, so called ‘crescendo TIAs’. The most dramatic example is ‘capsular warning syndrome’, with escalating attacks leading to impending and disabling lacunar stroke - which is almost inevitable unless prevention measures are taken such as antiplatelets and blood pressure control. However, recurrent TIA-like episodes (i.e. sudden, focal, negative symptoms only) can also be due to other causes, for example blood overlying the cortex, and imaging is necessary.
Other causes of paroxysmal symptomsBeyond the ‘big three’ there are many, many other causes of transient neurological symptoms. They are a big part of what neurologists spend time assessing. It is impossible to list them all, but important categories are below. Some of these feature triggers, such as specific movements.
Hypoperfusion produces transient ischaemia. It may be due to limited inflow due to hypertension or a narrow vessel, or alternatively a steal phenomena - in which blood is shunted away from the target site.
i) Limited Inflow
This may globally affect the brain during hypotension arising on posture change or after eating, causing light headedness or syncope, sometimes after a delay - including several minutes. An unusual phenomenon is ‘limb-shaking TIA’ with single arm shaking due to a narrow carotid - which can mimic focal motor seizures.
ii)Steal phenomena
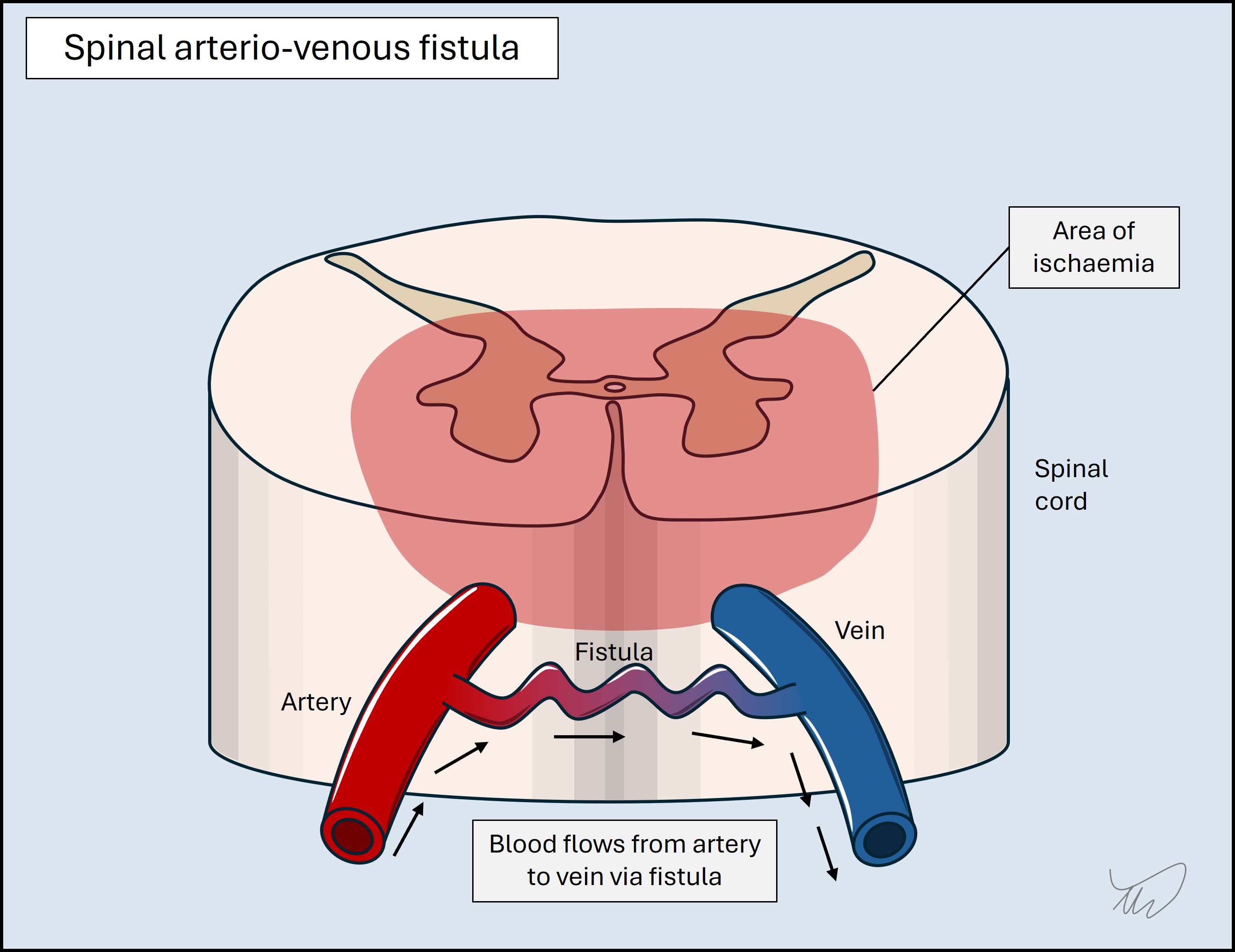
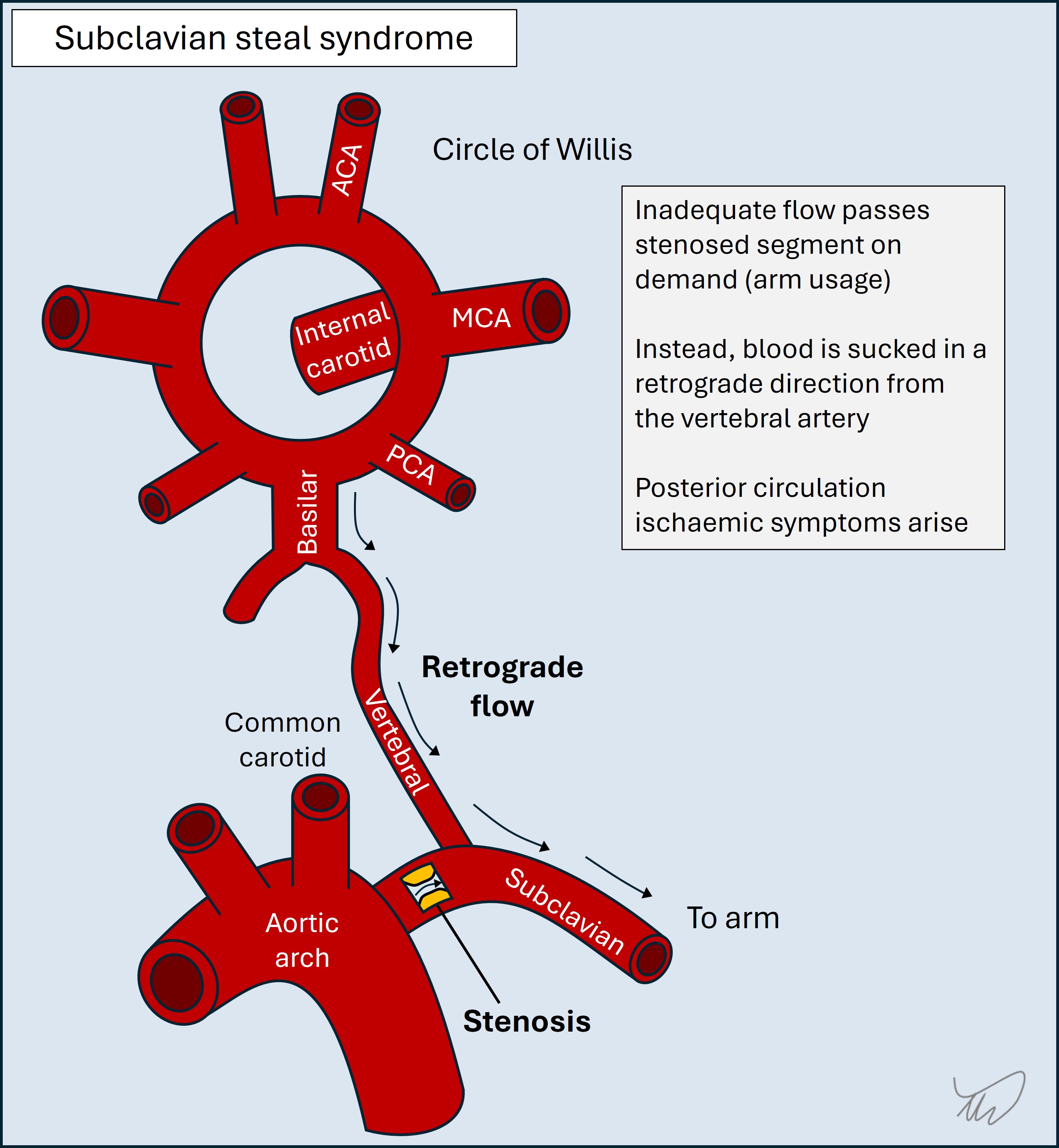
These arise when blood is either diverted into a vein (an arteriovenous fistula, AVF), bypassing the target, or is sucked into a collateral artery, with retrograde ‘steal’ from the artery supplying the now-hypoperfused target. AVFs in the spine are more active during standing and exertion, with episodic leg weakness improving on resting; steal is part of the mechanism, as well as venous hypertension. Subclavian steal syndrome is when subclavian narrowing (proximal to the vertebral artery origin) limits availability of blood to the arm on arm usage, so intracranial blood is sucked backward down the vertebral artery - leading to posterior circulation hypoperfusion.
Fluid flow may be obstructed within a channel. A common example is benign paroxysmal positional vertigo (BPPV), with recurrent episodes of vertigo on head movement due to crystals blocking the vestibular canals, which is debilitating yet easily treated. CSF in the ventricles can also be obstructed due to a mobile mass such as a colloid cyst, causing headache and collapse - a condition to never miss.
Compression of structures may only occur on dynamic movements. This is most common with peripheral nerves, especially the ulnar nerve at the elbow, causing tingling and sometimes involuntary movements and spasms during elbow flexion - for example while driving or leaning on the elbow. It can also occur with radiculopathy, for example during neck movement. We can confirm this during the exam using provocative manoeuvres (e.g Phalen, Spurling and straight leg raise tests).
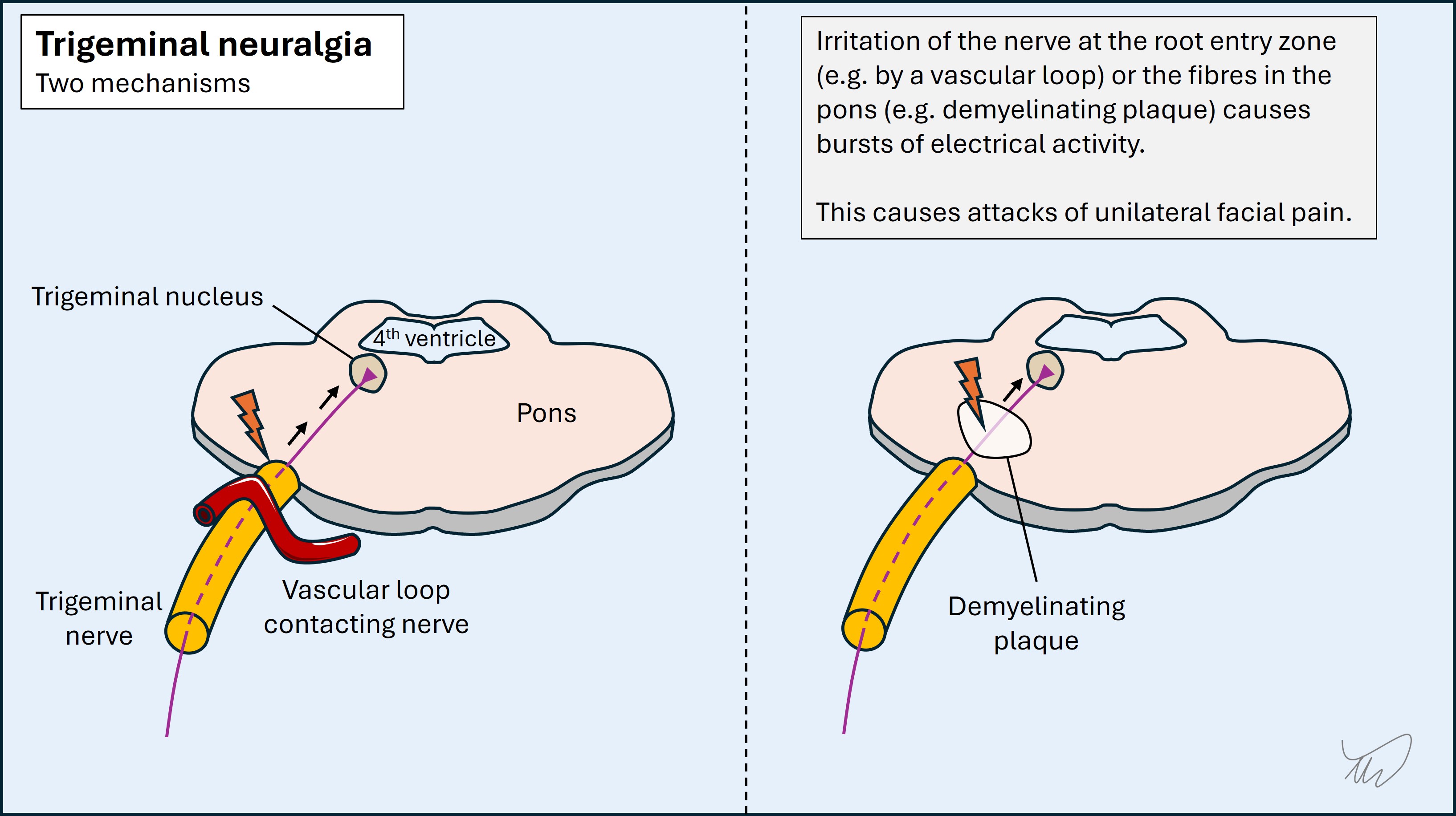
Electrical discharges in the spine or cranial nerve roots (ephaptic transmission) may cause pain, abnormal sensations, or involuntary movements. This can arise following parenchymal damage, particularly brainstem or spine demyelination, as well as nerve root compression at the brainstem by a blood vessel or a mass. Attacks can sometimes be triggered, for example by touching skin. Trigeminal neuralgia is the most notorious, with bursts of agonising hemifacial pain. Lhermitte’s phenomenon, electric shocks radiating down the neck and into the arms on neck flexion, is another example.
Some metabolic or toxic disturbances can cause transient symptoms if the problem peaks (or troughs in the case of metabolic deficiencies) and then wears off. Hypoglycaemia can mimic TIA/stroke with focal deficits such as weakness or dysarthria. Drugs (including prescribed medicines) can cause transient symptoms as the level peaks, for example ataxia, vertigo and diplopia with anti-seizure medicines, or involuntary movements on various stimulants of abuse.
There are many genetic disorders that produce paroxysmal symptoms such as stiffness, paralysis, ataxia, or involuntary movements - typically due to mutations in genes encoding ion channels. Examples include myotonia, periodic paralysis, episodic ataxia and paroxysmal dyskinesia. Onset is generally in childhood, teens or young adulthood. They do not present for the first time in older people.
Other clues - temporality, triggers, relievers and additional symptomsAdditional characteristics provide clues to the type of lesion/pathology, including diurnal variation, triggering or relieving factors, and other associated symptoms.
Diurnal variation is seen in some conditions. Myasthenia (neuromuscular junction) is worse in the evenings as are some movement disorders, particularly dopa-responsive dystonia, and headaches due to intracranial hypotension. In contrast some disorders often manifest at night, for example cluster headache or frontal lobe seizures.
Demyelination-related symptoms are often worse in heat, including on exercise, during a shower, or amid heatwaves - known as Uhthoff’s phenomenon.
Alcohol and caffeine may worsen or ameliorate symptoms of numerous disorders. Physiological tremors are made worse by caffeine, while alcohol can reduce the effects of essential tremor, as do GABA-ergic medications.
Stress has worsening effects on many neurological disorders - it’s difficult to think of any that improve with it - so the presence of recent stress is not very helpful in terms of thinking about one condition or another. It is a common error to assume symptoms are functional when they follow stress, but so can seizures and strokes. Stress can also be a coincidence, and if you look for it, most patients can identify some source of recent stress - so do not be misled by it!
Some aetiologies may have accompanying symptoms which are worth identifying.
Infection is often accompanied by symptoms such as fever, sweats, rigors, muscle aches and lethargy. They are not always present, and are not specific to infection and can be seen in other types of illness (for example cancer or drug toxicity) - but are important clues to consider. However, the situation can be complicated, as some symptoms are due to inflammation either accompanying infection (para-infectious, for example Bell’s palsy) or following it (post-infectious, most famously Guillain-Barré syndrome).
Cancer can be accompanied by weight loss, lethargy and loss of appetite (anorexia) - i.e. cachectic symptoms. This is certainly true of cancers that metastasise to the nervous system, although primary nervous system tumours often lack these additional clues. These symptoms can also be seen in systemic inflammatory disorders such as sarcoidosis and vasculitis. Some chronic infections cause them, too; this is why tuberculosis used to be called ‘consumption’.
Where, what… and who?The primary way we assess for a lesion site and location should be the history itself - not who the patient is and their risk factors for a given disease, including demographic factors and comorbidities. These do have value, but they are supplementary - adding or reducing weight for certain disorders. Place most weight on the symptoms, not who has them.
Do neurology long enough and you’ll realise that anyone can have anything, and errors are made by discounting possibilities because the patient doesn’t seem like the typical demographic to have a given problem - for example, considering stroke to be unlikely due to young age or lack of (known) risk factors - or the other way round: over-diagnosing stroke/TIA in older people who may have alternative causes.
Among the most problematic is the bias against women, with symptoms often dismissed as anxiety or functional that would be investigated differentially in men. This is made worse by the tendency of medicine to teach symptoms according to how they are usually experienced in men - some are quite different in women, but our training under-prepares us for this reality.
Base your diagnosis on ‘Where?’ and ‘What?’, rather than ‘Who?’ - but reserve the latter for the formulation, when demographic features can increase or reduce the odds of a given condition. Think of them as risk factors rather than direct diagnostic indicators.
There are some general statements below - none are ‘hard rules’, but reasonable principles.
General considerations - patient groups with elevated risks of certain disordersAn older person with a sudden-onset focal neurological disorder and who has multiple vascular risk factors is likely to have had a stroke, although other differentials exist.
If an older person presents with an acute headache, while it may be a migraine, there is a higher probability that they have an underlying serious condition, so investigation may be warranted - whereas a 20 year old with the same headache may more reliably be given reassurance without further testing, assuming a reassuring history and no 'red flags' such as sudden-onset or focal features.
In a young woman with focal symptoms which came on over hours-days and persisted, demyelination is an important consideration - though far from the only one. Functional disorders are much commoner in young women than young men, but should be diagnosed carefully and based on clear functional features, and never on sex.
A person with immunocompromise (e.g. HIV, cancer, transplant, or immunosuppressive medication) is at risk of infection. For a given presentation such as headache or focal deficits, the threshold to investigate for infectious causes is low, and we should test for atypical infections - for example toxoplasmosis.
In someone with cancer, or a past history of it, consider nervous system complications of the cancer - even years later. This is particularly true of certain cancers which are prone to invading the nervous system, such as breast and lung cancer.
In a pregnant woman, various disorders are more common, including cerebral venous sinus thrombosis (CVST). While many headaches occur in pregnancy for benign reasons, CVST is a consideration, likewise with new-onset focal neurological problems and seizures, so our threshold to look for this condition is lower.
Avoiding the trap of not investigating due to lack of (known) risk factorsNot only should we be careful about letting risk factors overly bias us towards specific conditions - we should also not discount other conditions due to the lack of known risk factors.
Some people are not known to have risk factors they later turn out to have. A person not known to have risk factors for cerebral venous sinus thrombosis (CVT) may well have both CVST and a risk factor for it. We should find the CVST then work out why they have it, rather than discounting it on the basis of what we don’t know.
It's not uncommon for a stroke to manifest in someone with previously-unknown risk factors. This is especially true in the young, with important aetiologies including arterial dissection and cardiac anomalies - generally unknown before the stroke. The mistake is assuming stroke is not possible due to young age and lack of known risks. If the presentation suggests stroke, test for it - then find out why it happened.
Paraneoplastic syndromes are an interesting example, where we usually find the remote neurological consequences of the cancer (such as encephalitis or neuropathy) before the cancer itself - sometimes months or years earlier. The syndrome is identified, and we then hunt for the tumour. Indeed the specific syndrome influences which tumours we look for - for example breast or gynaecological cancer in cerebellar degeneration. We certainly do not discount the possibility of paraneoplasia in a patient not known to have neoplasia!
EthnicityEthnicity influences predisposition to various disorders, so it does have some role in our diagnostic thinking - but it should not bias us, particularly into discounting the possibility of a disease altogether.
Ethnicity doesn’t heavily influence the primary ‘where and what?’, but it may come into consideration after, fine-tuning our suspicion for particular diseases. It does not however influence whether or not we think someone has a disease from one of the general categories (e.g. vascular or inflammatory). It should never bias us towards not taking a patient seriously on the basis of their ethnic background. Unfortunately such biases are quite heavily conditioned into medical and societal thought, so it is important to try to become conscious of such prejudices.
If we suspect myelitis, MS may be less likely than other disorders in people who are not white, and depending on the ethnic group other causes might include neuromyelitis optica, Behçet's disease or sarcoidosis. We might look for additional clinical evidence for these (e.g. mucosal ulcers).
Stroke aetiologies may be influenced by ethnicity, for example with intracranial atheroma being commoner in Asian people.
Ethnicity is especially relevant for genetic disorders, with genetic variations associated with individual diseases being more prevalent in certain ethnic groups. There are countless examples, but having some awareness of these can help us recognise them - but only if the features are suggestive, and never on ethnic grounds alone.
SexSex has a lot of relevance, particularly around treatment considerations, and may add some weight to diagnostic thinking - but again, should not overly.
Many diseases are more prevalent in one sex, but most are not 100% exclusive. Men get cluster headache more commonly than women, and vice versa for MS, but women also get cluster headaches and men get MS.
Functional neurological disorders, myasthaenia, migraine and subarachnoid haemorrhage are all commoner in women than men but plenty of men also suffer with them. The reverse is true, too - with many women suffering diseases commoner in males such as motor neuron disease and Parkinson's.
A few examples are shown below - the proportions give a sense of the scale of these differences. True rates can be challenging to identify given age is a risk factor for many of these and there are higher numbers of elderly women than men.
Commoner in women than menFor the most part, these numbers are not high enough to make it appropriate to discount a diagnosis in a sex group that has it less commonly. Having some awareness of the epidemiology helps us make sensible decisions. Dismissing the possibility of a disease in a man because it is three times commoner in women is not sensible.
However, some conditions are so heavily associated with one sex that we should be cautious diagnosing them in the other. Idiopathic intracranial hypertension (IIH) is a likely explanation for headache and papilloedema in a woman with normal brain imaging and high-pressure CSF with normal constituents. In a man with the same BMI, symptoms, scan and CSF, it is unlikely - so we should consider alternative causes that can mimic it, such as sleep apnoea or jugular vein thrombosis. Women can have IIH mimics too, but we are more likely to go looking for these in men as IIH is quite rare in males.
Moving on...The history provides nearly all the information needed. We should be able to localise the problem in the nervous system, then consider timing to help us think about what the cause is - and factor in a few important health and demographic details carefully to modify probabilities of certain considerations. We should have a good sense of the likely diagnosis (or a shortlist of ‘suspects’) in our minds as we move on to the exam.
Exam